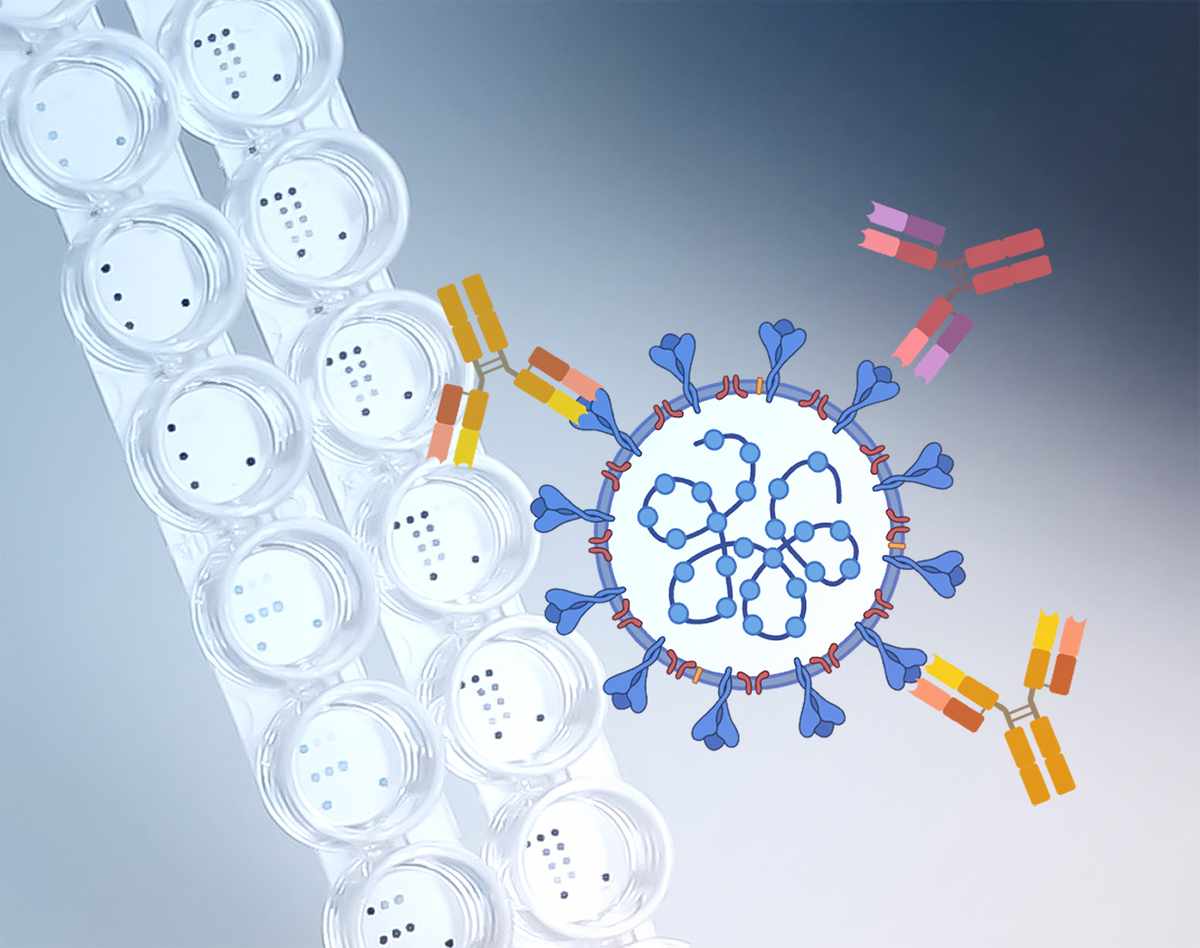
To defend itself against infectious diseases and foreign substances, the human immune system produces special proteins against the pathogen – so-called antibodies. However, these antibodies are not directed against the entire pathogen, instead they always bind to very specific molecular structures of the intruder via a lock-and-key principle. These structures are also known as "antigens". InfectoGnostics scientists have now developed a test platform in the RESISTOVAC project, on which dozens of such antigens can be applied. A single drop of the patient's blood can thus be used to test in parallel for several antibodies against common and new infectious diseases.
The central focus of the development was initially a more precise screening of the immune reaction to the coronavirus. A so-called protein microarray was used for this: This is a chip a few millimetres in size on which various antigens are applied as capture molecules. If antibodies from the patient's blood are brought together with the matching antigens on the chip, a specially developed method leads to a staining of corresponding spots on the microarray – a targeted antibody must therefore have been present in the blood.
In order to validate the reliability of this detection, the InfectoGnostics researchers applied different SARS-CoV-2 antigens to the microarray and tested them against real samples from Covid-19 patients from Thuringia: "We wanted to detect the relatively late-formed IgG antibodies – a kind of long-term memory of the immune system against components of a pathogen. To detect them, we applied 18 antigen combinations for the coronavirus as capture molecules," explains Sindy Burgold-Voigt, first author of the study and doctoral student at the Leibniz-IPHT.
Cooperation of several partners from the InfectoGnostics research campus
The microarray was designed by Leibniz-IPHT scientists from the "Optical-Molecular Diagnostics and Systems Technology" working group headed by Prof. Ralf Ehricht, together with developers from fzmb GmbH in Bad Langensalza. The microarray was finally manufactured on the basis of the "Inter-Array" system from the fzmb GmbH. Another company partner of InfectoGnostics, Virion\Serion GmbH, also provided antigens for Corona and other pathogens.
For antigen referencing and validation of the test, the Jena University Hospital contributed biobank samples and data from their 2020 Corona study "CoNAN" in Neustadt am Rennsteig. The village in northern Thuringia was completely quarantined at the beginning of the pandemic and systematically recorded by JUH researchers in an epidemiological study.
Immune systems react extremely individually to coronaviruses
In their evaluation, the scientists found that the immune response varies greatly from individual to individual: for example, some patients had only developed antibodies against a protein that envelops the genetic material of the virus – the nucleocapsid. Other patients, however, had antibodies against the so-called spike protein – the pointed structures with which the virus attaches itself to human cells. Still others had matching antibodies against both corona antigens.
The clever combination of different antigens on a single test can thus be of great importance for the understanding of antibody immune defence and the work on new vaccines, explains Sindy Burgold-Voigt: "For the further development and efficiency analyses of vaccinations, it is extremely important that research has a diagnostic tool with which one can get a quick overview of the immune response."
Microarray for all STIKO vaccinations could provide clues to vaccination gaps
In addition to various surface structures of the coronavirus, three antigens from the pathogens for diphtheria, measles and tetanus, to which vaccinated individuals typically react, were also put on the test. Here, a corresponding antibody response was successfully detected in vaccinated individuals. "We were thus able to show that we can flexibly extend the test and detect different antibodies during a single test in the patient's blood. In the future, we could therefore compile a microarray for all vaccinations recommended by the STIKO, with which we could screen for possible vaccination gaps quickly and cheaply," explains Sindy Burgold-Voigt.
The test development was supported by the Federal Ministry of Education and Research as part of the InfectoGnostics lead project RESISTOVAC, which aims to develop POC tests to determine immune status and bacterial resistance factors. In addition, the study was funded by the Free State of Thuringia and co-financed by European Union funds under the European Social Fund (ESF).
Publication: Burgold-Voigt S, Müller E, Zopf D, Monecke S, Braun SD, Frankenfeld K, Kiehntopf M, Weis S, Schumacher T, Pletz MW, Ehricht R; CoNAN Study Group. Development of a new antigen-based microarray platform for screening and detection of human IgG antibodies against SARS-CoV-2. Sci Rep. 2022 May 16;12(1):8067. DOI: 10.1038/s41598-022-10823-7.